

August 2014
IN THIS ISSUE:
![]()
- 3D printing now available in hospital
- First installation of new CT technology in clinical setting
- New 'wayfinding' tools will help patients navigate Clinical Center
- Palmore named Hospital Epidemiologist; Henderson recognized for service
- Early communication with FDA can reduce clinical drug development time
- NIH 'knocks out hunger' on campus, in community
- Upcoming events on rare Erdheim-Chester disease will feature NIH researchers, patients
- NIH Sibling Day empowers 'super sibs'
- Upcoming Events
Print this Issue ![]() (431 KB)
(431 KB)
ABOUT CC NEWS:
![]()
Published monthly by the Office of Communications and Media Relations. News, article ideas, calendar events, letters, and photographs are welcome. Submissions may be edited.
Clinical Center News
National Institutes of Health
Building 10, 10 Center Drive
Room 6-2551,
Bethesda, MD 20892-1504
Tel: 301-594-5789
Fax: 301-402-0244
Molly.hooven@nih.gov
QUICK LINKS:
![]()
3D printing now available in hospital
 |
The 3D printer at the NIH Library in the Clinical Center uses a bioplastic derived from corn. |
In June, the NIH Library at the Clinical Center opened a new space called the Technology Sandbox to highlight technology-based projects at NIH.
One of those technologies is a 3D printer, which can create hollow and solid 3D models by building up thin layers of material into a predefined shape.
"[This] is an opportunity for our staff and our customers NIH staff to become familiar with this new technology and find useful ways to apply it to their work and research at NIH," said MaShana Davis, an information architect with the library.
The printer is available on a first-come, first-served basis; no reservation is required. Users need to complete a 30-minute orientation, which is held weekly at the library. Seating is limited to 10 attendees at a time with registration available through the Center for Information Technology.
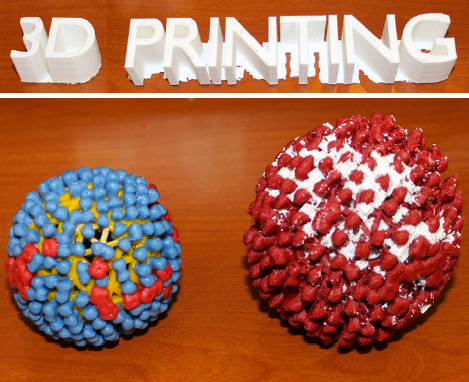 |
The text and HIV molecules are examples of what can be printed in the Sandbox. |
"The printer itself is self-service. Library staff is on hand to provide assistance if needed," said Verma Walker, a biomedical librarian with the library.
The printer is available for all staff. Printed models must be applicable to NIH work or research. Printing can take upwards of 20 hours to complete.
The library is offering this 3D printing service for free until Aug. 29. Users will be asked to complete an evaluation once they have finished printing their model. This trial period will help assess the demand and potential cost.
"We definitely want NIH staff to take advantage of the Technology Sandbox," Davis said. "We see this as an opportunity for our customers to explore different technologies not readily available at their institutes or centers, to collaborate with colleagues they would not usually work with and to discover other library services that might help in their work or research at NIH."
The Technology Sandbox includes:
Online resources
Want to have your project featured in the Technology Sandbox? Call 301- 496-1080 or email NIHLibraryTechnologySandboxProject@mail.nih.gov |
First installation of new CT technology in clinical setting
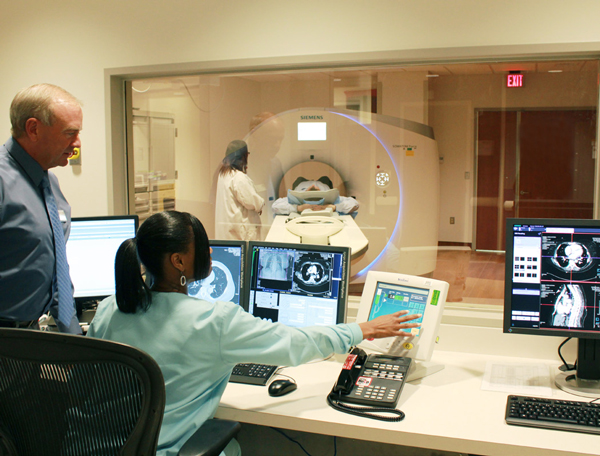 |
In addition to a lower radiation dose, the scanner will also provide better quality scans and faster run times. The scanner evaluates the entire chest, abdomen and pelvis in one second. |
An estimated 62 million Computerized Tomography (CT) scans are conducted in the US every year [disclaimer], including the more than 25,000 here at the Clinical Center. Some patients require CT scans once a week during critical phases of their therapy trials. With patient safety continuously at the forefront of their minds, the Radiology Department installed the latest generation, high-speed CT scanner that will use the lowest radiation levels available.
The equipment, just approved by the Food and Drug Administration, puts the Clinical Center at the forefront of patient care with the first clinical installation in the US. This follows the research testing installations at the Mayo Clinic in Rochester, Minn. and the Medical University of South Carolina in Charleston, SC.
"This new CT scanner represents another 'first' for the NIH Clinical Center," said Dr. David Bluemke, director of the Radiology and Imaging Sciences Department. "It reinforces the commitment of the NIH to the highest level of safety and technology for our patients and research subjects."
CT scans are an effective diagnostic tool as they create detailed anatomical images and can help physicians enhance treatment and avoid unneeded medical procedures. However, they use ionizing radiation, a known human carcinogen, which poses a potential drawback for patient health. In addition, the changing body types of Americans, an irregular heartbeat, impaired kidney function, age and/or advanced illness have all provided challenges to using the technology.
Despite radiation concerns, CT is often the most effective and rapid method to assess the lungs and diagnose abnormalities of the abdominal and pelvic organs and bones. The radiology department has addressed these concerns under the direction of Dr. Les Folio, staff clinician and lead radiologist for CT. Folio assembled a dose reduction team in 2010 to reduce radiation exposures in half with the new scanner.
Folio outlined the advantages of the high-tech equipment.
"Although we already scan with as low of radiation doses as most any hospital, we will now be able to provide even lower doses," Folio said.
A National Cancer Institute study also has shown that low-dose CT can reduce lung cancer mortality rates by 20% compared to standard x-ray procedures. According to Siemens, the manufacturer of the equipment, the scanner reduces radiation doses up to 50% compared to similar CT systems.
The equipment was installed in the newly-renovated CT area in July. Working with top architects, the radiology department strived for a welcoming yet efficient layout. The environment included effective sight lines and airport like status screens where staff can have at-a-glance knowledge of workflow.
Radiology technologists and physicists have completed performance and radiation safety checks to ensure the scanner was ready for use; followed by safety inspections. CT scans are an important tool in patient diagnosis and treatment. The Clinical Center's efforts to put patient safety first are central to providing safe and effective clinical studies.
Palmore named Hospital Epidemiologist; Henderson recognized for service
 |
Dr. Tara Palmore (center), Clinical Center Hospital Epidemiologist, with members of the Hospital Epidemiology Service, Angela Michelin (left), infection control consultant, and Brooke Decker, clinical fellow. |
Dr. Tara Palmore will serve as the new Hospital Epidemiologist at the Clinical Center. Palmore was Deputy Hospital Epidemiologist for the past seven years, working alongside the previous lead, Dr. David Henderson. Henderson will continue as Deputy Director for Clinical Care and Associate Director for Hospital Epidemiology and Quality Improvement.
"She's the future of the program for us. She's incredibly intelligent, has great personal skills and all the things you need," said Henderson, who was the first person to be named Hospital Epidemiologist at the Clinical Center in 1979.
Henderson hopes to watch Palmore and future hospital epidemiologists enable 'health care to become the equivalent of a high reliability organization [such as] air traffic control, the computer industry and the nuclear power industry — all zero defects, zero tolerance places."
In the senior leadership position, Palmore will manage a staff of infection control consultants; collaborate with the public health department; determine if infections were acquired in the hospital and look for trends among positive cultures. She will work with the Hospital Epidemiology Service which manages hospital acquired infections while preparing for those developing across the world. The Middle East Respiratory Syndrome (MERS), Severe Acute Respiratory Syndrome (SARS) and pandemic influenza are just a few health conditions for which they continue to track and prepare guidance.
Palmore first started collaborating with Henderson in 2005. She says with a smile that she didn't realize at the time she was being 'reeled in' to becoming a health care epidemiologist. Just two years later she was invited to serve on the Hospital Epidemiology Service.
"From that moment on I really enjoyed working in this field," she said. "I hope to continue not only improving our methods of prevention hospital infections and our effectiveness in doing so but also increasing the science base for infection control interventions. It's not just the content of the job it's the people that I get to work with that make me look forward to coming in every day."
Recently, Palmore and Henderson along with Drs. Julie Segre and Evan Snitkin earned a Samuel J. Heyman Service to America Medal and the prestigious title of Federal Employees of the Year in 2013 from the Partnership for Public. They were honored for their success in identifying, tracking and eliminating an antibiotic-resistant bacterial outbreak in the Clinical Center with the use of DNA sequencing.
"You don't get a blue ribbon for doing your job well when there are no outbreaks," Palmore said. "The more quiet things are and the less people think about us the better we're doing our job."
With the support from the Clinical Center administration, the Hospital Epidemiology Service will continue to make advances to better protect those at the heart of all the research — the people.
Palmore will continue to serve as Program Director for the NIAID Infectious Diseases Training Program, Chair of the Clinical Center's Hospital Infections Committee and member of the DHHS Prevention and Implementation Working Group of the Hospital Acquired Infections Steering Committee.
Early communication with FDA can reduce clinical drug development time
A meeting with the Food and Drug Administration (FDA) may decrease the time it takes for clinical development of drugs by an average of about three years.
In 2013, FDA researchers studied the clinical development times of all drugs approved between 2010 and 2012. On average, it takes 10 to 15 years to develop a drug. When a clinical investigator of the drug held a pre-investigational new drug (IND) meeting with the FDA before beginning clinical trials, the FDA researchers found that the average development was three to six years shorter.
"We think early communication can make a big difference regarding quality and efficiency," said Dr. Anne Pariser, Associate Director for Rare Diseases, Office of New Drugs, Rare Diseases Program at FDA's Center for Drug Evaluation and Research.
A clinical investigator must submit an IND application to the FDA before he or she can test a new drug in human subjects. The application typically will include information about the drug's non-clinical toxicology profile and any safety information available from prior human administration, drug formulation and characterization, proposed dosage, and the proposed clinical protocol and monitoring plan. The FDA wants to ensure that clinical trial participants are protected from unnecessary risk and reviews the IND application mainly focusing on safety for first-in-human and early phase clinical trials.
Prior to the application submission, the investigator can request a meeting with the FDA to ask for advice on clinical trial design and to learn about necessary IND-enabling elements, including pre-clinical pharmacology and toxicology.
"Any investigator can request a meeting with the FDA. However, these early meetings are particularly important for the development of drugs for rare diseases," said Pariser.
To schedule a pre-IND meeting, an investigator must submit a written request to the FDA. Should the meeting request be granted, FDA will try to schedule the meeting within 60 days of receipt of the written request. The clinical investigator should also submit the background package for the meeting, as well as questions to be addressed at least 4 weeks before the meeting. Pariser recommended scheduling the meeting prior to conducting animal toxicity studies.
Dr. Pariser co-chaired a Joint Task Force with Dr. Juan Lertora, Clinical Pharmacology Program director in the Office of Clinical Research Training and Medical Education at the NIH Clinical Center. The task force encourages and facilitates early interactions with FDA regulatory staff.
View our February 2014 CC News article on IND applications. Read FDA guidance ![]() (97 KB).
(97 KB).
NIH 'knocks out hunger' on campus, in community
 |
Corey Welcher, John Veitch, Kian Memarzadeh, Nancy McLean-Cooper and Rodelyne Augustin help Feds Feed Families 'Fill the Truck' outside of the Clinical Center. |
The NIH donated more than 5,000 pounds of canned and boxed food and beverages to the Feds Feed Families food drive since June 1. The drive will continue through August 27. In addition to food items, donations also included hygiene products such as soap, toothpaste, tissues and shampoo.
New this year, in an effort to directly serve the community, a portion of the NIH donations will be provided to The Edmond J. Safra Family Lodge and The Children's Inn [disclaimer]. Both facilities experience lulls in donations during the summer and the Feds Feed Families program helps fill that need. The remainder will be distributed by the Capital Area Food Bank [disclaimer] to communities in Maryland, the District of Columbia and Virginia.
"Two can Tuesdays" and "Fill the Truck" promotions helped encourage donations. There are 17 donation sites on campus, including 3 in the Clinical Center, and 24 at nearby sites. NIH offices in Frederick and Baltimore, Md. and Montana and North Carolina are also participating.
Purchase food online to be donated and learn more.
Upcoming events on rare Erdheim-Chester disease will feature NIH researchers, patients
The 2nd Annual International Erdheim-Chester Disease (ECD) Medical Symposium will bring distinguished researchers together to discuss the rare condition that, without successful treatment, can lead to organ failure and death. The Symposium will take place September 18 in Building 60.
ECD is a very rare form of non-Langerhans cell histiocytosis in which abnormal white blood cells start to rapidly grow in number and encase or infiltrate parts of the body such as the bones, kidneys, skin, and brain. Signs and symptoms, as well as disease course, depend on the specific location and extent of involvement. The disease can be debilitating with patients having problems with memory or needing to rely on wheelchairs, walkers and feeding assistance. There are no treatments and research continues to find potential causes.
The symposium will be held by the NIH National Human Genome Research Institute (NHGRI), the NIH Office of Rare Disease Research and the ECD Global Alliance [disclaimer]. With more than 45 patients enrolled at the Clinical Center, NHGRI has the largest ECD natural history study in the U.S.
Thomas Caron, who's been in the study for three years, had his third visit to the Clinical Center in April 2014. He met other participants and learned more about ECD.
"It presents differently in every single person, so it's not like it's a cookie cutter disease," Caron said. "I told my wife when I got diagnosed 7 years ago that I'm going to do whatever I can do to help out so somebody else doesn't have to go through what we went through."
Caron hopes to join about a hundred other patients and families from the NIH and around the world at a separate gathering, sponsored by the ECD Global Alliance, September 19 and 20 at a Bethesda hotel. Attendees will meet other patients with ECD and their families as well as leading NIH and external researchers from the U.S., Europe, Israel and Australia who will discuss patient trials, diagnosis, treatment standards and other topics presented just a few days before at the symposium.
"It's important to go especially because caregivers can have their own support group. My wife is my caregiver and she has nobody who she can talk to regarding the illness," said Caron. "If we can make the symposium, she will be able to talk with other caregivers. I can find out more information from other ECD patients; how they're doing, what their involvement is and then learn all sorts of information from all the medical providers."
The first International ECD Medical Symposium was held in October 2013 in California. Erik Ray, another patient in the study who came to the Clinical Center in April 2014, talked about attending that event.
"I stopped by to not feel alone so much in this disease," said Ray, noting that the disease affects each person differently depending on which organ is affected. "After talking to a few people, I realized that we're all alone. This is different for all of us. The research they're gathering right now can help the next guy. In that sense, I don't mind helping the future."
View an ECD Global Alliance video [disclaimer] with patient's stories and the ongoing work in the Clinical Center. For more details on the events email estradaverasji@mail.nih.gov.
NIH Sibling Day empowers 'super sibs'
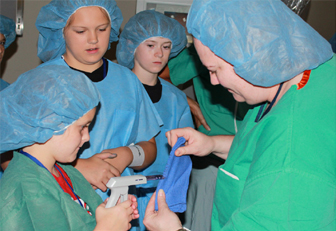 |  |
Brothers and sisters of pediatric patients attended Sibling Day July 15 for a behind the scenes look at medical research. Staff from the Clinical Center, The Children's Inn [disclaimer], National Cancer Institute and National Institute of Mental Health worked together to organize hands-on activities at the Inn and in the operating room, mock MRI scanner and lab. They also participated in therapeutic games and art activities. The 'super sibs' got a surprise visit from Batman who helped recognize them for the important role they play as part of the health care team. See local press coverage from Fox 5 [disclaimer] and CBS Radio [disclaimer].
Upcoming Events
All events listed below will be streamed and archived.
Clinical Center Grand Rounds Lecture
Contemporary Issues in Graduate Medical Education
New Models for Large Prospective Studies Involving Genomics
August 13, 2014; Noon - 1:00 p.m.
Lipsett Amphitheater
Presented by Teri Manolio, MD, PhD, NHGRI
Clinical Center Grand Rounds Lecture
Contemporary Issues in Graduate Medical Education
Challenges in Health Care Delivery: From the Iron Triangle to the Three Aims
August 20, 2014; Noon - 1:00 p.m.
Lipsett Amphitheater
Presented by Piyush K. Agarwal, MD, NCI
Clinical Center Grand Rounds Lecture
Contemporary Issues in Graduate Medical Education
Better Handoffs, Safer Care: Improving Transitions of Care to Reduce Medical Errors and Improve Patient Safety
August 27, 2014; Noon - 1:00 p.m.
Lipsett Amphitheater
Presented by Amy J. Starmer, MD, Harvard Medical School
Twenty Years of Allogeneic Stem Cell Transplantation at NIH
Day 1: Scientific Program- Advances in the Practice of Stem Cell Transplantation
September 11, 2014; 8:00 a.m. 5:00 p.m.
Masur Auditorium
The event will describe the current state of the art of allogeneic stem cell transplantation as illustrated by two decades of clinical research at the Clinical Center.
Day 2: Survivorship Program- Patient and Caregiver Perspective
September 12, 2014; Noon - 4:00 p.m.
FAES Terrace
The celebratory day, which is open to patients and caregivers, will cover the benefits and challenges faced by survivors and their families following treatment.
View more details and register online.
The National Symphony Orchestra
September 17, 2014; 1:30 p.m. 2:30 p.m.
Hatfield Building, Atrium
A special performance, co-presented by the Foundation for Advanced Education in the Sciences and the Clinical Center.
NOTE: PDF documents require the free Adobe Reader.
 The information on this page is archived and provided for reference purposes only.
The information on this page is archived and provided for reference purposes only.