Clinical Center News
Role reversal for NINDS fellow, CRTP alum
 |
(from left): Dr. Michael Dimyan, a neuro-rehabilitation fellow with NINDS’ Human Cortical Physiology Section of the Medical Neurology Branch and CRTP participant Erick Tarula, a fourth year medical student from Charles Drew/UCLA, look at a computer image of Tarula’s brain.
|
by Nicole Martino
“See one, do one, teach one.” A motto within medicine expressing the obligation to teach those a step behind, it’s the goal of the Clinical Research Training Program (CRTP) that places research-minded medical and dental students in mentored clinical or translational research projects in a field matching their personal interests and goals.
Eight years ago Dr. Michael Dimyan, now a neuro-rehabilitation fellow with the human cortical physiology section in the NINDS, participated in CRTP. He returned to NIH two years ago and now helps to mentor current CRTP fellow Erick Tarula, a fourth-year medical student from Charles Drew University of Medicine and Science. He spent the first two years of his program at the University of California-Los Angeles’s (UCLA) David Geffen School of Medicine in Southern California.
According to Dimyan, CRTP presents an exceptional opportunity for medical students to interact directly with fellows. “For medical students training at other hospitals, there is usually little exposure to fellows, who primarily teach senior residents.” This program is different. Medical students work with fellows at a time when fellows really begin to definite themselves career-wise.
“Observing the fellows—who themselves are learning new techniques, navigating unwritten rules, and demonstrating that instead of an instruction booklet there are multiple paths to reach your goals—helps medical students clarify their understanding of themselves, what they want, and how they want to get it,” he said.
Dimyan credits his return to NIH to the positive experience he had as a medical student in the CRTP. He describes the year as extremely inspiring and influential. So much so that after leaving NIH, he kept in touch with both mentors Dr. Mark Hallett, chief of the NINDS human motor control section, and Dr. Takashi Hanakawa, currently a professor of cortical function disorders at the National Center of Neurology and Psychiatry in Tokyo. Dimyan’s project during his 2000- 2001 CRTP year focused on imaging brain activity during motor planning,imagery, and movement.
Also influential was Hallett’s ability to bridge the gap between being a scientist and a clinician. “He is a rare person in still being an outstanding clinician and an amazing scientist as well. There are very few people who can do both,” he said. “Observing subtle aspects of behavior and movement, which are the products of the nervous system, is what makes a great neurological diagnostician. Then as a clinical researcher, the challenge is to use those observations to address what might be a subtle, yet scientifically critical question,” Dimyan said. CRTP demonstrated a way for Dimyan to mix the clinical and scientific aspects of neuroscience, which helped shape him and his career.
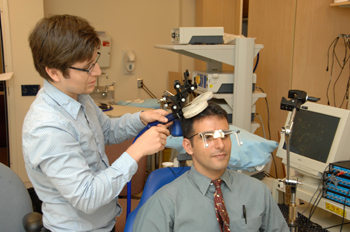 |
(from left): CRTP participant Erick Tarula, a fourth year medical student from Charles Drew/UCLA, and Dr. Michael Dimyan, a neuro-rehabilitation fellow with NINDS’ Human Cortical Physiology Section of the Medical Neurology Branch, demonstrate a device that marks the precise position of parts of the brain in space. The infrared markers and coils placed over a patient’s skull register on the computer over brain images previously captured during an MRI to create a real-time image of the brain in three dimensions.
|
Now, eight years later, Tarula is also benefiting from the program’s emphasis on mentoring around both clinical research skills and professional development. Dimyan and Tarula work together in Dr. Leonardo Cohen’s laboratory studying brain functioning through electromagnetic stimulation. Tarula’s CRTP project focuses on the cerebellum’s role in motor learning using transcranial magnetic stimulation.
With about 90 students interviewing for 30 CRTP slots, Tarula said he feels lucky to be selected and to have Dimyan as one of his mentors. “Michael really took me under his wing, showed me the ropes, and helped me develop my own project. He has helped mature both my clinical and scientific skills things I need to become a successful clinician-scientist,” Tarula said.
Smiling and laughing as they walk down the Clinical Center’s halls, Dimyan and Tarula look more like old friends then mentor and student. “Neither of us really had a lot of family in the American medical system. It has been interesting sharing the culture of medicine, helping him avoid certain things, or improve on things I would have liked to do better,” Dimyan said. In addition to their mutual interest in neuroscience and background growing up in California, the two men share a passion for music and politics. “I used to be a DJ on my college’s radio station. Now Erick introduces me to new music,” said Dimyan.
Tarula is already continuing the medical community’s tradition of mentoring. He works with students at his former high school and spoke recently at the National Network of Latin American Medical Students conference, encouraging others to go into clinical research. Throughout his career, Tarula hopes to use his developing
clinical research skills to identify and address previously unanswered neurological problems.
When asked about his future goals, Dimyan said that neuroscience, which in the past has mostly offered diagnoses but few treatments, has recently blossomed with new approaches to patient rehabilitation. “I don’t want to let go of stroke patients after they are stabilized. I want to develop the tools and methods therapists can use to achieve greater levels of rehabilitation. I feel confident that with a brain-centered approach, we can improve rehabilitation,” he said. Dimyan also just ended his tenure as clinical cochair of the Fellows Committee, which he describes as a “collegial and energizing experience.”
As a mentor, Dimyan has encouraged Tarula to be confident in the work that he has put into his CRTP project and to project that confidence to his interactions with medical colleagues and patients. “Your patients depend on you to be confident especially in neurology where you see a lot of bad disease that robs memories, personality, and a lot of what makes you human.”
Editor’s Note: Nicole Martino, a public and community health major at the University of Maryland, College Park, interned this summer with the Office of Communications, Public Liaison and Patient Recruitment.
Back to Top
Clinical Center greets new chief director of radiology
 |
Dr. David Bluemke, new CC chief radiologist
|
Dr. David Bluemke has been named the Clinical Center’s director of Radiology and Imaging Sciences. He also has been awarded tenured scientist status at NIH.
In announcing the appointment, Dr. John I. Gallin, CC director, said, “Dr. Bluemke’s breadth of experience and expertise will be an extraordinary asset to NIH’s imaging sciences programs.”
Bluemke, who will hold joint appointments at NHLBI and NIDDK, had been clinical director of the MRI division at Johns Hopkins and professor of radiology and medicine at the Johns Hopkins University School of Medicine.
Noting that imaging research is by nature multi-disciplinary, Bluemke said his goals for his new role are to develop and enable a broad range of research initiatives, clinical expertise, and training.
Bluemke, an American Heart Association fellow, conducts research on cardiovascular disease and its complications and seeks to better understand how subclinical disease can be detected with newly developed imaging technologies, described, and tracked over time.
His group at Johns Hopkins was the first to describe the use of myocardial delayed enhancement in arrhythmogenic right ventricular dysplasia (ARVD). He is the world-recognized imaging expert in this extraordinarily difficult-to-diagnose condition and was the principal investigator of the MRI reading center for the US ARVD multi-center trial. His group was also one of the first to demonstrate a method for multi-station MR angiography for peripheral vascular disease and to use MRI for direct intravascular, ultra-high resolution evaluation of atherosclerotic plaque in humans. His expertise in imaging also includes oncologic disease and the evaluation of cancer with MRI.
He was a lead author on four multicenter clinical trials evaluating the use of novel contrast for MRI for oncologic applications, the largest multi-study of MRI in the evaluation of breast cancer, and a multi-center trial in cardiovascular disease. He is currently studying cardiovascular disease with MRI in the Epidemiology
of Diabetes Interventions and Complications trial involving more than 1,000 type 1 diabetic patients and the Multi-Ethnic Study of Atherosclerosis study of 6,800 individuals.
To date, Dr. Bluemke has authored more than 260 peer-reviewed publications and 27 book chapters and monographs. He sits on the editorial boards of several journals: Radiology, the Journal of Computed Axial Tomography, Journal of Magnetic Resonance Imaging, Applied Radiology, and the International Journal of Cardiovascular Imaging.
He is a member of the board of trustees for the Internal Society of Magnetic Resonance in Medicine (ISMRM), board member of the North American Society of Cardiovascular Imaging, current chair of the American College of Radiology (ACR) Cardiac Accreditation Committee, past program chair of the ISMRM Cardiac MR Study Group, and has been appointed to the AHA Council on Radiology and Intervention, the ACR Committee on Standards for Body MRI, and for Standards and Accreditation for Body and Neuroradiology Magnetic Resonance.
His most recent awards include an outstanding teacher award from ISMRM in cardiovascular imaging, the Journal of the American College of Cardiology elite reviewer award, two Executive Council awards and a silver medal from the American Roentgen Ray Society, and Cum Laude and contrast agent awards from the Society of Computed Body Tomography and Magnetic Resonance. In 2007 he was named among the top 10 radiologists by Medical Imaging and among America’s Best Doctors by Castle Connolly Medical Ltd.
He earned his medical degree at the University of Chicago’s Pritzker School of Medicine as part of the medical-scientist training program and his PhD from the University of Chicago’s Department of Biophysics and Theoretical Biology, where he studied the evaluation of three-dimensional macromolecules using advanced computer modeling techniques. He holds an undergraduate degree in chemical engineering from the University of Wisconsin-Madison and a master’s in business from Johns Hopkins University. He was a fellow in cross-sectional imaging in diagnostic radiology at Johns Hopkins School of Medicine and completed residency training at the Johns Hopkins Hospital.
Back to Top
‘A child will be accepted because you helped them with surgery’
 |
(from left): Martha “Marti” Shepherd and Margaret Schmidt with some of their anesthesiology equipment at the Clinical Center
|
Before doctors and nurses on medical missions to developing countries can complete their surgical procedures, a very necessary step must be taken care of: anesthesiology.
Martha “Marti” Shepherd and Margaret Schmidt, two of the CC’s certified registered nurse anesthetists, have traveled the globe to help share their expertise.
Shepherd volunteered for a month in the Philippines in 2005 with the Philippine Medical Association of DC and the Philippine Nurses’ Association of DC.
Schmidt volunteers with Mercy Ships, a global charity that delivers free health care and community services in developing countries. She has volunteered eight times, most recently in January 2007, to work on the Mercy Ship Anastasis in West Africa. Her work with Mercy Ships has also taken her to Senegal, Ghana, South Africa, Guinea, Benin, Gambia, and Sierra Leone.
Working in a small space on the ship, overcoming language barriers, and dealing with stressful, life-threatening cases, Schmidt brings her compassionate demeanor to these patients to help give them a brighter future. Patients are underweight or malnourished. Despite the challenges, getting through long and difficult surgeries on a ship that may turn and cause the anesthesia cart to roll away unexpectedly and seeing patients doing well afterwards makes it worthwhile.
Back to Top
New CC News editor
|

|
CC News editor Maggie McGuire
|
Maggie McGuire joined the CC as editor of CCNews on July 21.
In her previous post at NHGRI, McGuire wrote and edited content for the institute’s Web site and the NIH Record.
She holds an undergraduate degree in print journalism from the University of Maryland, College Park.
McGuire has interned at The Sun, Baltimore, and The Times Union, Albany,NY, and worked as a marketing associate at Smithbucklin in Washington, DC.
Back to Top
BTRIS status to be presented at town hall
by Michelle Anderson
After six intensive months of design and development, a proof-of-concept demonstration of BTRIS – the Biomedical Translational Research Information System – will be presented at a September 16 town hall meeting at 1 p.m. in Lipsett Amphitheater.
“BTRIS is intended to provide researchers with easy access to clinical data derived from multiple sources, including the clinical information systems at the Clinical Center, for their on-going trials,” says Dr. James Cimino, chief of the Laboratory for Informatics Development and BTRIS project director. He notes that the system also allows investigators to search across data sets to answer new questions and shape fresh ideas for future studies.
At the upcoming town hall, Dr. Cimino hopes to help researchers visualize the full potential of BTRIS, obtain feedback and kick-off a “demonstration period, so that when BTRIS is implemented next year, it meets the needs of all those who will use it.”
For those unable to attend, the meeting will also be available on videocast at http://videocast.nih.gov.
Back to Top
Protocol tests type 2 diabetes medication to treat severe asthma
 |
NHLBI’s Dr. Stewart Levine, technician Clara Jolly, and patient and fellow Jonathan Lam demonstrate the pulmonary function tests that trial participants will receive to monitor the progress of their asthma.
|
Two Japanese patients with type 2 diabetes and asthma sparked the idea. They started taking pioglitazone hydrochloride (HCI), a medication used to treat type 2 diabetes, and noticed that their asthma also improved.
Coincidence? There is no published data in humans on the topic, so it’s impossible to know until research can move beyond anecdotal evidence. Dr. Stewart Levine, principal investigator with NHLBI’s Pulmonary and Vascular Critical Care Medicine Branch, launched one of the first randomized, placebo controlled, double-blind pilot clinical trials in patients with allergic asthma to find out whether pioglitazone HCI (Actos) is effective for treating patients with asthma who do not respond to standard therapy.
Pioglitazone belongs to a class of medications called thiazolidinediones, which have been shown to have anti inflammatory effects. These medications have been used to improve asthma in pre-clinical studies, so there is compelling evidence that this approach might be useful for asthmatic patients.
According to Levine, 22 million people in the US have asthma, and approximately 10 percent of those patients have severe cases. The mild to moderate treatments don’t work for them, so Levine’s goal is to find new treatments for people with moderate to severe asthma. Often already using inhalers, these patients’ next options would be oral corticosteroids or injectable anti-IgE therapy. Levine hopes that about 50 asthma patients between 18 and 75 years of age who have had asthma for at least one year and whose symptoms are not well controlled with current treatments will choose to enroll in his study.
The study has three phases. First, candidates are screened with blood and urine tests, breathing tests, an allergy skin test, chest x-ray, electrocardiogram, and echocardiogram. Participants are given a device to measure and record lung function and asthma symptoms at home for four weeks before starting the study medication. Lung function is also measured at clinic visits before and after inhaling a bronchodilator medicine and participants are admitted to the hospital for fiberoptic bronchoscopy and lavage—a test to examine the lung’s airways.
In phase two, participants are randomly selected to receive either pioglitazone or placebo, a look-alike pill with no active ingredient, once a day for 10 weeks. They return to the clinic after two weeks to repeat the tests done in the first phase and to monitor any reactions to the study drug or placebo. If there are no problems, the amount of medication is increased and they return for follow-up evaluations every two weeks for eight weeks. Pulmonary function tests and bronchoscopy and lavage are repeated after 10 weeks on medication.
In the last phase, patients return for follow up one month after stopping the medication or placebo to monitor their asthma. Because pioglitazone is an already FDA approved medication commonly prescribed for type 2 diabetes, about seven million people have already taken it. According to Levine, the main side effects are weight gain, edema, and anemia. Long-term use of this class of medication has been associated with worsening heart failure in diabetic patients who use insulin, as well as bone fractures, so patients will be monitored with bone density assessments.
If the results from Levine’s pilot study look promising, he hopes to open it up in a few years to patients with severe asthma at multiple locations. According to Levine, there is an important unmet need for the development of new treatment options for patients with severe asthma. “If pioglitazone or other medications could be shown to be effective for the treatment of severe asthma, this would represent a wonderful new option for these patients.”
For more information, call 1-800-411-1222 or TTY 1-866-411-1010.
Back to Top
Novel cancer vaccine developed from stem cell transplant donors
by Jenny Haliski
How do you improve on existing therapies to make them more effective and improve the prognosis for patients? That puzzle has fascinated a multi-disciplinary group of NIH researchers together with extramural colleagues for several years.
Clinical Center Bedside-to-Bench awards (see sidebar, below) in 2003 and 2004 seeded investigations that have
now led to introducing a new cancer vaccine for the first time in human patients. It’s also one of the first attempts to use healthy stem cell transplant donors to develop a cancer vaccine. The vaccine represents a last hope for the patients who receive it in this phase1/2 pilot trial, but researchers hope that if successful, similar vaccines may someday be incorporated into initial cancer treatments.
The problem: 20 percent of children with acute lymphoblastic leukemia (ALL) the most common pediatric malignancy accounting for about a quarter of all childhood cancers die from the disease. Even worse, approximately half of adult patients with ALL die from it.
Allogeneic stem cell transplantation (BMT) is an immunotherapeutic approach to treating ALL. Part of the success of BMT is a “graft-versus-leukemia (GVL)” effect—where the donor immune system eradicates residual leukemia be-potent in ALL as it is in other hematologic malignancies.
According to clinical principal investigator Dr. Alan Wayne, the overriding goal of this project was to bring together expertise to develop a new cancer vaccine strategy to enhance the GVL effect after BMT for ALL and other hematologic malignancies. Wayne is also clinical director and head of the hematologic diseases section in NCI’s Pediatric Oncology Branch and Center for Cancer Research.
 |
(from left, standing): Dr. Elaine Jaffe, NCI; Dr. Terry Fry, NCI; Dr. Rodger Kurlander, CC; Dr. David Stroncek, CC; Kelly Richards, NCI; Hua Zhang, NCI; Dr. Mary Alice Stetler-Stevenson, NCI; Dr. Ronald Gress, NCI; and Dr. Mark Raffeld, NCI. (from left, seated): Dr. Alan Wayne, NCI; and Dr. Crystal Mackall, NCI.
|
This novel cancer vaccine trial is designed to treat children and adults with hematologic malignancies that have recurred or persisted despite a previous allogeneic stem cell transplant. This represents one of the first attempts in humans to utilize cells obtained from healthy stem cell transplant donors to develop cancer vaccines. In the past the process of creating cancer vaccines typically used the patient’s own blood, bone marrow, or tissue. Wayne’s process synthesizes products taken from individuals other than the patient. The trial also involves the National Marrow Donor Program (NMDP) and makes both related and unrelated donor/recipient transplant pairs eligible to participate.
One common approach to treating relapsed leukemia after a BMT is to ask the healthy transplant donor to give lymphocytes for infusion into the patient. In another type of leukemia this donor lymphocyte infusion (DLI) has an 80 per-cent cure rate, but for ALL, it is less than 10 percent. One reason is that ALL cells are less “visible” to the immune system than other types of leukemia. Wayne and colleagues wanted to develop a vaccine that would make ALL cells more visible to lymphocytes. To do this, they created a vaccine made up of dendritic cells, which present foreign proteins to the lymphocytes directing their attack.
As part of this trial, at the same time the donor’s lymphocytes are collected for DLI, another type of white blood cells are also donated. These are then cultured and matured in vitro into dendritic cells by the CC’s Department of Transfusion Medicine (DTM) Cell Processing Section staff. In one of the Bedside-to-Bench aspects of this project, team investigators modified techniques developed at the NIH in the 1990s for use with autologous dendritic cell cancer vaccines and adapted these for cells obtained from healthy BMT donors. The project is part of the legacy of Charles Carter, who led the DTM cell processing section’s developmental efforts on this project, which were completed around the time of his death in 2006.
Patients on this trial receive two small vaccine injections from the cultured donor cells every other week. Once each month they also receive standard donor lymphocyte infusions. The entire course of outpatient treatment occurs over a three-month period with visits to the CC day hospital.
The dendritic cell vaccine targets WT1, a gene involved in pediatric kidney tumors that is also linked to childhood and adult cancers, including ALL. The vaccine utilizes three different WT1 peptides, which are pieces of proteins. Wayne’s team also uses a new type of peptide technology called protein transduction domains (PTD) designed to make the vaccine more immunogenic. PTD peptides were synthesized linked to the WT1 peptides for use in the vaccine. Project investigators hope that the use of allogeneic dendritic cells and PTD technology after BMT will make this vaccine approach among the most potent forms of immunotherapy available. NIH researchers have partnered with Dr. David Loeb, an authority on WT1, at Johns Hopkins University.
A Bedside-to-Bench award in 2003 and 2004 with support from the NCI, NHLBI, CC, and NIH Office of Rare Diseases, funded pre-clinical studies in project investigator laboratories and synthesis of clinical grade peptides for used in the clinical trial. The trial was approved in January by the FDA and the NCI and NMDP institutional review boards.
The first patient received the investigational vaccine in the spring and other patients have been screened at the CC for possible future participation. Wayne and colleagues hope to test the vaccine in 12 individuals before moving on to the next phase of study. Two of the team’s long-term goals are to modify the vaccine to include all immune system (HLA) types. The current vaccine only targets HLA type A2, which 40 percent of the population has—and to use the full WT1 protein, rather than just three peptides.
“We are excited about all of this expertise and multiple technological advances converging on a clinical need: better therapies for individuals whose cancer relapses after a stem cell transplant,” Wayne said. “It would be unprecedented to have a high cure rate in this group of patients. We are targeting those at highest risk.” Although Wayne said the current trial is designed to test whether the vaccine is safe and effective for patients who have already relapsed, he could envision a time when the vaccine is given prophylactically as part of the initial BMT. This would be ideal because it is best to cure disease as early as possible and intervene when the immune system is capable of rapidly responding. “It’s always a race between the disease and the treatment. The less disease patients have at the start of the race, the better off they are,” Wayne said.
Back to Top
Clinical fellows’ meet and greet at welcome reception
 |
Clinical Center Director Dr. John I. Gallin (right) meets Dr. Jonathan Berkowitz (left), a clinical fellow with NCI's Medical Oncology Branch, at the reception for new clinical fellows.
|
New clinical fellows met and mingled with Clinical Center and NIH leaders at the July 9 welcome reception. It’s an annual tradition at the conclusion of the CC-sponsored orientation program that brings people from different countries, medical specialties, and experiences to come together.
The fellows, who participate in a number of clinical training programs at NIH, including many accredited through the Accreditation Council for Graduate Medical Education, typically spend two to five years working in their own clinical specialties with senior clinical investigators.
The orientation and reception are just the beginning of a fellow’s time at NIH. The first year’s focus is clinical research and patient care at the Clinical Center. Subsequent training may involve clinical or basic research or a combination of both. Some of the training is offered at partner institutions such as National Naval Medical Center, Children’s National Medical Center, and Johns Hopkins University Hospital.
Back to Top
Clinical Center launches patient ambassador volunteer program
 |
(at left) Denise Ford, chief of hospitality services and patient ambassador program coordinator, and Courtney Duncan (at right), director of volunteer services and patient ambassador program co-coordinator, stand with one of the first groups of patient ambassadors at their orientation.
|
The Clinical Center will soon launch a new special volunteer program. Patient ambassadors, who will be recognizable in their burgundy jackets and nametags, will accompany patients between appointments within the hospital. Initial assignment areas will include transporting patients to radiology and surgery, moving medical records throughout the building, and taking the daily wheelchair inventory.
Approximately 70 patient ambassadors are already trained and committed to one year of serving at least four hours each week. As the program grows, Courtney Duncan, acting director of volunteer services in the CC’s Social Work Department, will recruit, interview, hire, and train new patient ambassadors. For information about the application process, see www.cc.nih.gov/about/ambassadors/.
Back to Top
Principles of Clinical Pharmacology course registration opens
The 2008-2009 CC Principles of Clinical Pharmacology course will begin on September 4 in Lipsett Amphitheater. The course will be held Thursdays from 6:30 p.m. to approximately 7:45 p.m. and will run through April 23, 2009. The registration deadline is August 22.
Course topics include pharmacokinetics, drug metabolism and transport, assessment of drug effects, drug therapy in special populations, and drug discovery and development.
Dr. Juan Lertora, director of the CC clinical pharmacology program, a member of the Office of Clinical Research Training and Medical Education (OCRTME), and course director since July 2006, said an outstanding faculty has been assembled to present the lectures, drawing from NIH scientific staff, FDA, the pharmaceutical industry, and many prestigious US academic institutions. “We have been very pleased with the great interest generated by this course,” said Dr. Frederick P. Ognibene, OCRTME director.
Registration is open to all at no cost, Principles of Clinical Pharmacology course registration opens unless the course is being taken for graduate credit through FAES as PHAR 500 I and PHAR 500 II. Contact FAES at 301-496-7976 for credit. Certificates will be awarded at the end of the course to all students attending 75 percent of the lectures.
Additional information, including online registration, is available by accessing www.cc.nih.gov/training/training/principles.html or by calling Donna Shields, clinical pharmacology program coordinator, at 301-435-6618.
Back to Top
CC employees honored with NIH Director’s Awards
 Four Clinical Center employees received NIH Director’s Awards in a July 21 ceremony at Natcher Auditorium.
Four Clinical Center employees received NIH Director’s Awards in a July 21 ceremony at Natcher Auditorium.
Clockwise from top left: Michael Alexander, hospitality services coordinator, accepts his award for creating a welcoming environment from NIH director Dr. Elias Zerhouni (right) with Dr. John I. Gallin, CC director; Dr. Bradford Wood, inerventional radiologist in the Diagnostic Radiology Department, received an award for leading the CC Imaging Science Program during a time of dramatic change; Joseph Hendery, chief of credentialing services, (also shown with Dr. Zerhouni and Dr. Gallin) was honored for providing high quality credentialing and privileging support; and Dr. Elizabeth Jones, deputy chief of radiology for clinical operations, received an award for her work with Dr. Wood in leading the CC Imaging Science Program during the program’s transition.
Back to Top
New clinical research protocols
The following new clinical research protocols were approved in June:
A Randomized, Double-blind, Placebo Controlled Trial (RCT) of Varenicline to Reduce Alcohol Consumption in Heavy Drinkers, 08-AA-0137, David W. Herion, MD, NIAAA
Phase II Study of Metastatic Cancer that Overexpresses p53 Using Lymphodepleting Conditioning Followed by Infusion of Anti-p53 TCR-Gene Engineered Lymphocytes and Dendritic Cell Vaccination, 08-C-0155, Steven A. Rosenberg, MD, NCI
Transfer of Autologous T Cells Transduced with the Anti-MART-1 F5 T Cell Receptor in High Risk Melanoma, 08-C-0162, Steven A. Rosenberg, MD, NCI
A Phase II, Multicenter, Open-Label Trial Evaluating the Activity and Tolerability of Romidepsin (Depsipeptide, FK228) in Progressive or Relapsed Peripheral T-cell Lymphoma Following Prior Systemic Therapy, 08-C-0164, Richard L. Piekarz, MD, NCI
An Open Label Pilot Study to Evaluate the Effect on the Immune System of Talactoferrin in Adults with Non-Small Cell Lung Cancer (NSCLC), 08-C-0166, Philip M. Arlen, MD, NCI
A Phase II Trial of Sunitinib in the Treatment of Recurrent Malignant Gliomas, 08-C-0168, Teri N. Kreisl, MD, NCI A New Method for Determining Gastric Acid Output Using a Wireless Capsule, 08-DK-0138, Stephen A. Wank, MD, NIDDK
Thyroid Hormones Homeostasis and Energy Metabolism Changes During Stimulation of Endogenously Secreted Bile Acids (BAs), 08-DK-0165, Francesco S. Celi, MD, NIDDK
Evaluation and Treatment Protocol for Potential Research Participants with Ocular Diseases, 08-EI-0169, Frederick L. Ferris III, MD, NEI
Screening of Subjects to Determine Eligibility to Safely Participate in Blood Disorders Studies, 08-H-0156, Matthew M. Hsieh, MD, NHLBI
VRC 015: A Phase I, Open-Label Clinical Trial to Evaluate the Safety, Tolerability and Immunogenicity of a Multiclade Recombinant HIV-1 Adenoviral Vector Vaccine, VRC-HIV ADV014-00-VP, in Uninfected Adults Randomized to Needle or Biojector Methods of Intr, 08-I-0171, Barney S. Graham, MD, NIAID
An Investigation to Determine Whether Levels of P11 Protein in Peripheral Blood Cells Correlate with Treatment Response to Citalopram in Patients with Major Depressive Disorder, 08-M-0150, Alan G. Mallinger, MD, NIMH
An Investigation of the Relationship Between Donepezil Enhanced REM Sleep, Sleep Architecture and Behavior in the Prepubertal Child with Autism, 08-M-0154, Ashura W. Buckley, MD, NIMH
PET Imaging of Peripheral Benzodiazepine Receptors Using [11C](R)-PK 11195 and [11C]PBR28, 08-M-0158, William C. Kreisl, MD, NIMH
The Role of Autoimmunity in Neurologic Complications of Celiac Disease, 08-N-0153, Ejaz A. Shamim, MD, NINDS
Noninvasive Language Mapping in Patients with Localization-Related Epilepsy, 08-N-0161, William H. Theodore, MD, NINDS
Back to Top
Upcoming Events
Clinical Center Grand Rounds for Clinical Fellows
August 6, 2008
Difficult Conversations: Evidence-Based Methods for Improving Outcomes
Elizabeth Rider, MSW, MD, FAAP
Director of Programs for Communication Skills, John D. Stoeckle Center for Primary Care Innovation, Massachusetts General Hospital;
Director of Academic Programs, Institute for Professionalism and Ethical Practice, Children's Hospital Boston;
Assistant Professor of Pediatrics, Harvard Medical School; Co-Chair, Medicine Academy, National Academies of Practice
August 13, 2008
Information Challenges in Clinical Research: Access, Rights, Explosions, & Other Hot Topics
Josh Duberman, BA, MLIS and Pamela Sieving, MA, MS, AHIP
NIH Medical Library
August 20, 2008
Who Goes First? Self-Experimentation in Medicine-Revisited
Lawrence K. Altman, MD
Senior Medical Correspondent and "The Doctor's World" columnist, New York Times
Clinical Professor, Medicine, New York University School of Medicine
August 27, 2008
Timing is Everything: Applying Circadian Rhythms Science to Duty Hours Requirements in Medical Training
Katherine M. Sharkey, MD, PhD
Assistant Professor, Warren Alpert Medical School of Brown University, Departments of Internal Medicine and Psychiatry and Human Behavior
Back to Top
This page last updated on 12/14/2017


 The information on this page is archived and provided for reference purposes only.
The information on this page is archived and provided for reference purposes only.